
SOLVED: A reaction mixture of 18.215 g Na2CO3 and 10.938 g HCl react according to the following balanced equation: Na2CO3 + 2HCl â†' 2NaCl + H2O + CO2 If 12.75 g of

The balanced equation is Na2CO3(s)+2HCl(aq)—>2NaCl(aq)+H2O(1)+CO2(g) The powder substance is 0.575The - Brainly.com
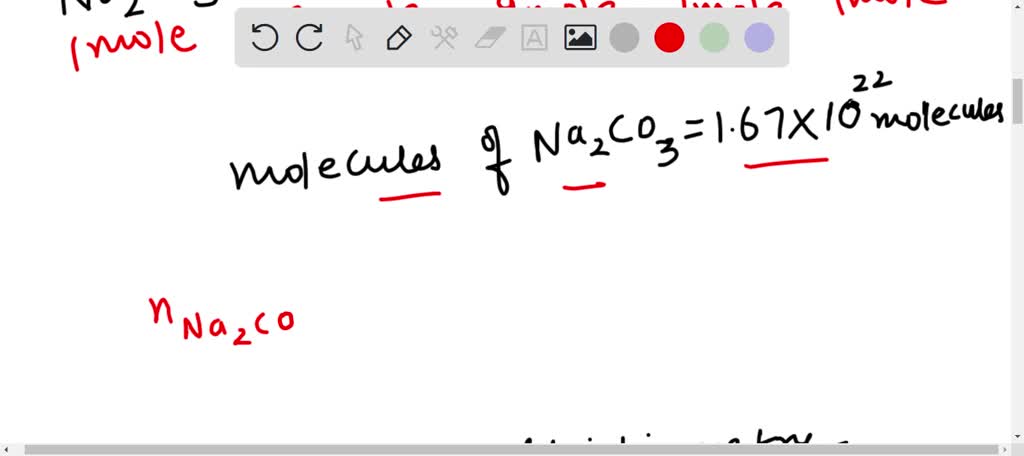
SOLVED: Na2CO3 + 2HCl ———> 2NaCl + CO2 + H2O How many moles of NaCl are produced from the reaction of 1.67 x 1022 molecules of Na2CO3 with excess HCl?
What are reasons for getting two different concentration values of Na2CO3 when it was titrated with HCL using Phenolphthalein and Methyl Orange? - Quora

SOLVED: A reaction mixture of 18.215 g Na2CO3 and 10.938 g HCl react according to the following balanced equation: Na2CO3 + 2HCl â†' 2NaCl + H2O + CO2 If 12.75 g of

10. Na2CO3 + 2HCL = 2NaCl + H2O + CO2 Какая массы воды выделилась, если в ходе реакции было - Школьные Знания.com

What are the tricks to learn reaction of sodium carbonate with hydrochloric acid? – The Unconditional Guru

SOLVED: Consider the reaction: Na2CO3 + 2HCl â†' 2NaCl + CO2 + H2O. A minimum of moles of sodium carbonate is needed to fully react with 750 g of HCl.

Welcome to Chem Zipper.com......: When 15 gm of NaCl and Na2CO3 is heated with dilute HCl, 2.5 gm of CO2 is evolved at NTP. Calculate percentage composition of the original mixture.

Question Video: Identifying the Observations of the Reaction between Hydrochloric Acid and Sodium Carbonate | Nagwa
24. What is the gram equivalent mass of Na2CO3 in a) Na2CO3 + 2HCl= 2NaCl +H2O b) Na2CO3 + HCl = Nacl + NaHCO3
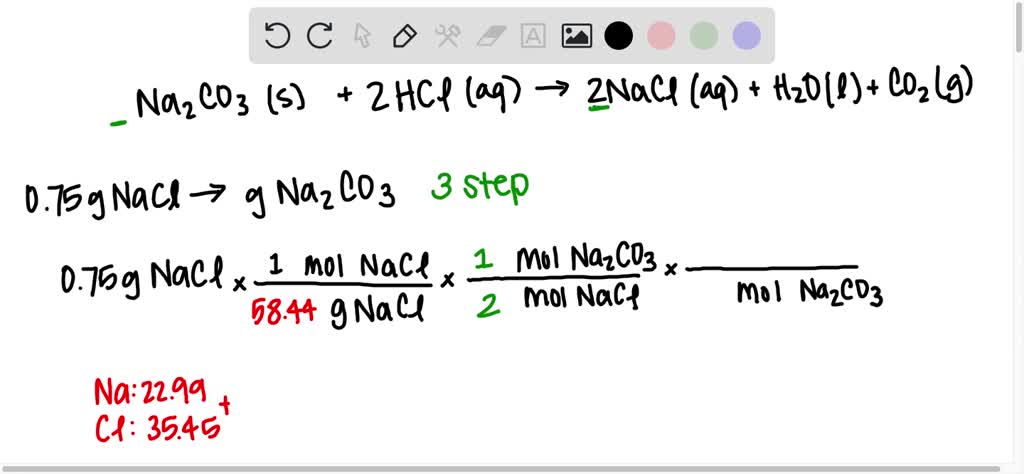
SOLVED: Na2CO3(s) + 2HCl(aq) â†' 2NaCl(aq) + H2O(l) + CO2(g) Using the balanced chemical reaction from step 1, calculate the mass of sodium carbonate (Na2CO3) that would be required to make 0.75

SOLVED: Hydrochloric acid (HCl) reacts with sodium carbonate (Na2CO3), forming sodium chloride (NaCl), water (H2O), and carbon dioxide (CO2). This equation is balanced as written: 2HCl(aq)+Na2CO3(aq)→2NaCl(aq)+H2O(l)+CO2(g) Part A What volume of 1.75









![Solved 0.32 M for 20 ml solution = [ Na2CO3] .9 Volume of | Chegg.com Solved 0.32 M for 20 ml solution = [ Na2CO3] .9 Volume of | Chegg.com](https://media.cheggcdn.com/study/ce6/ce696eaa-9751-4a1a-b5aa-f03d15d32ae3/image)