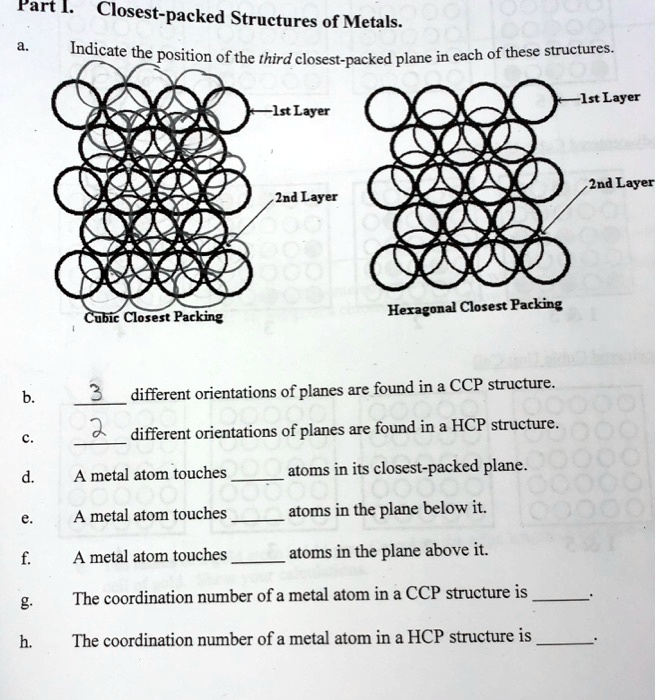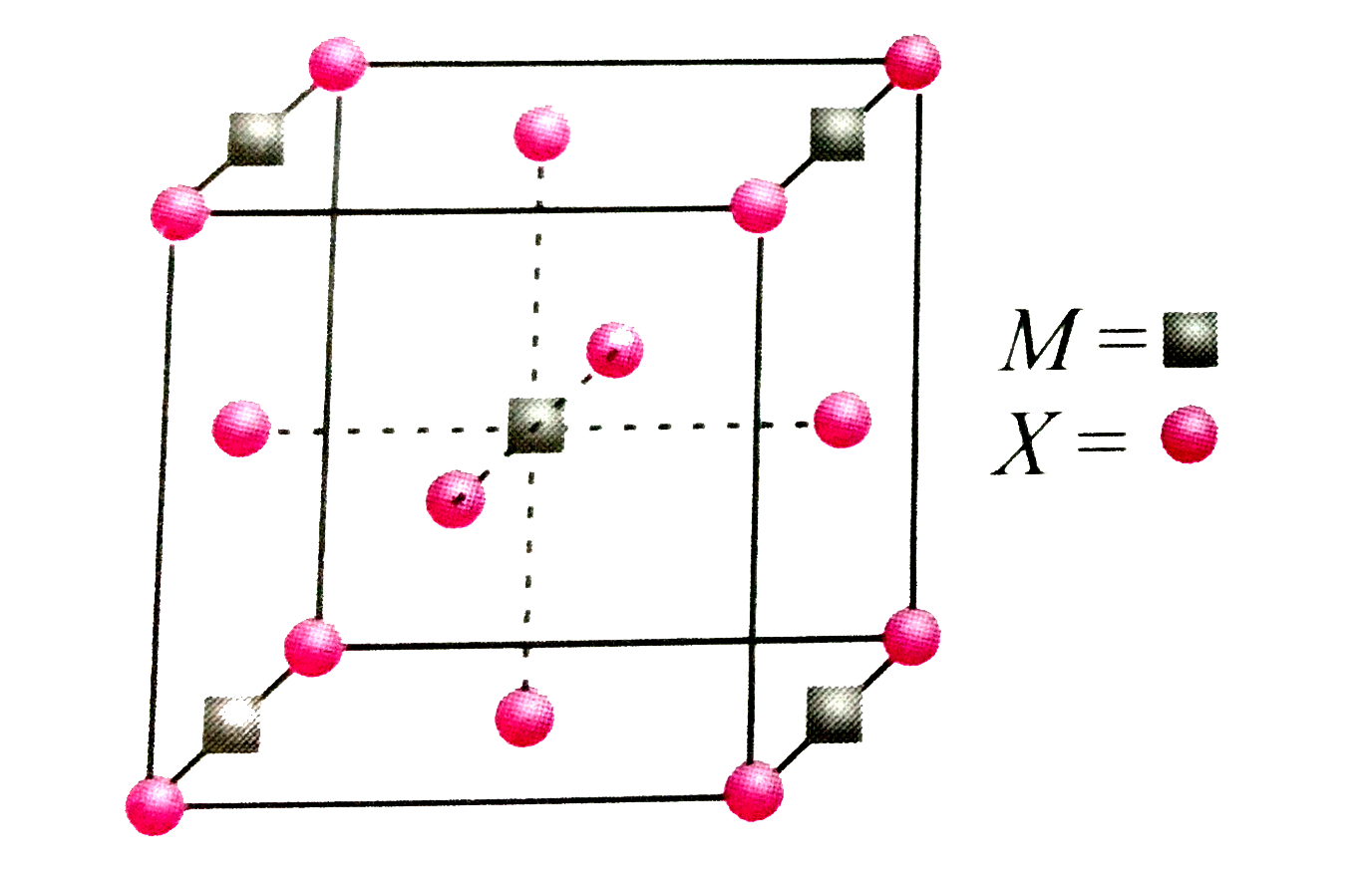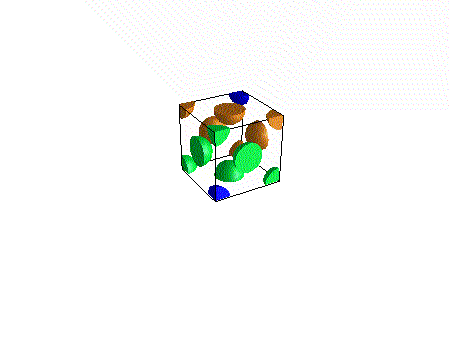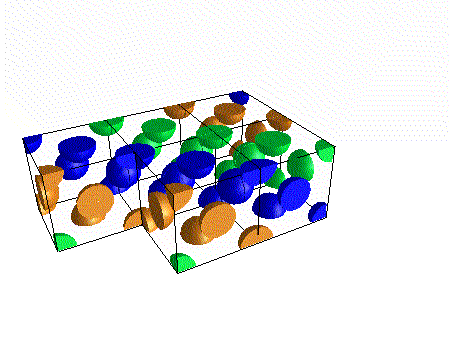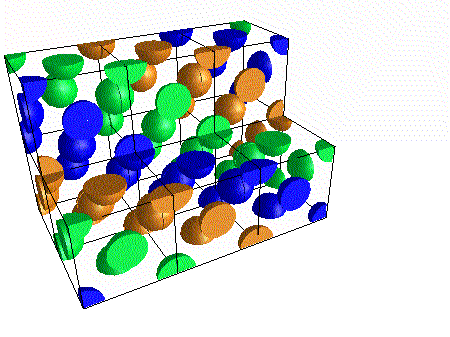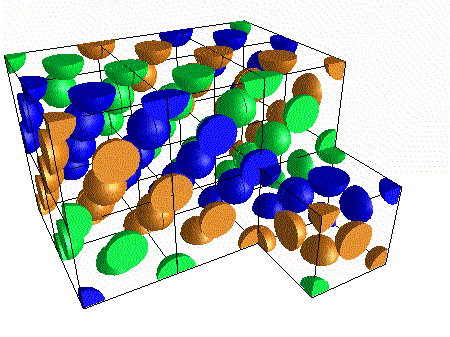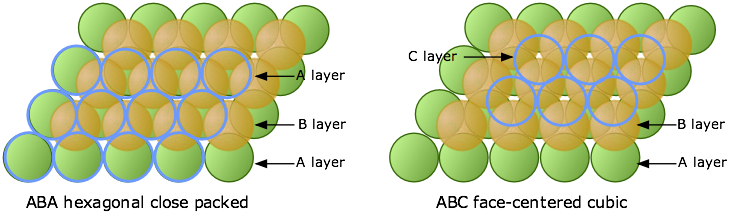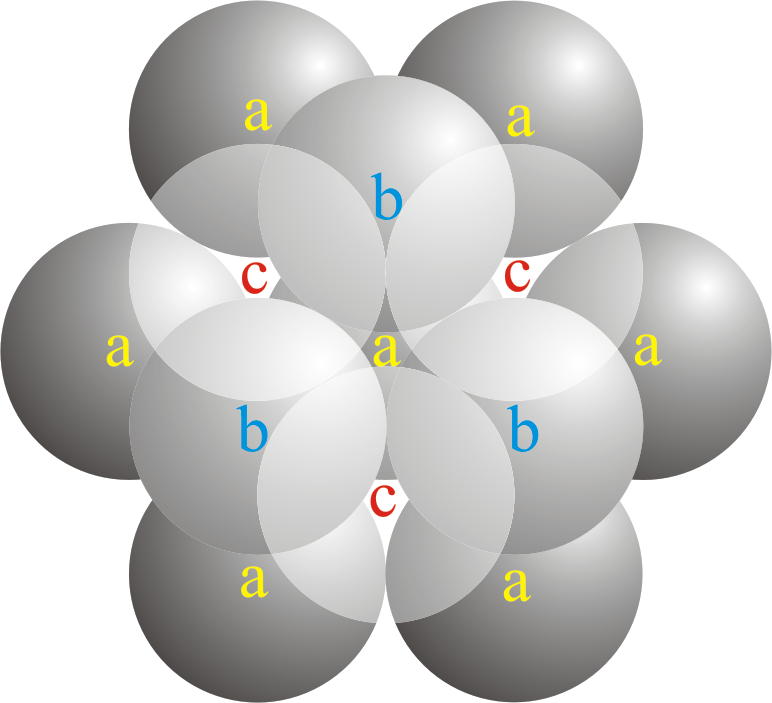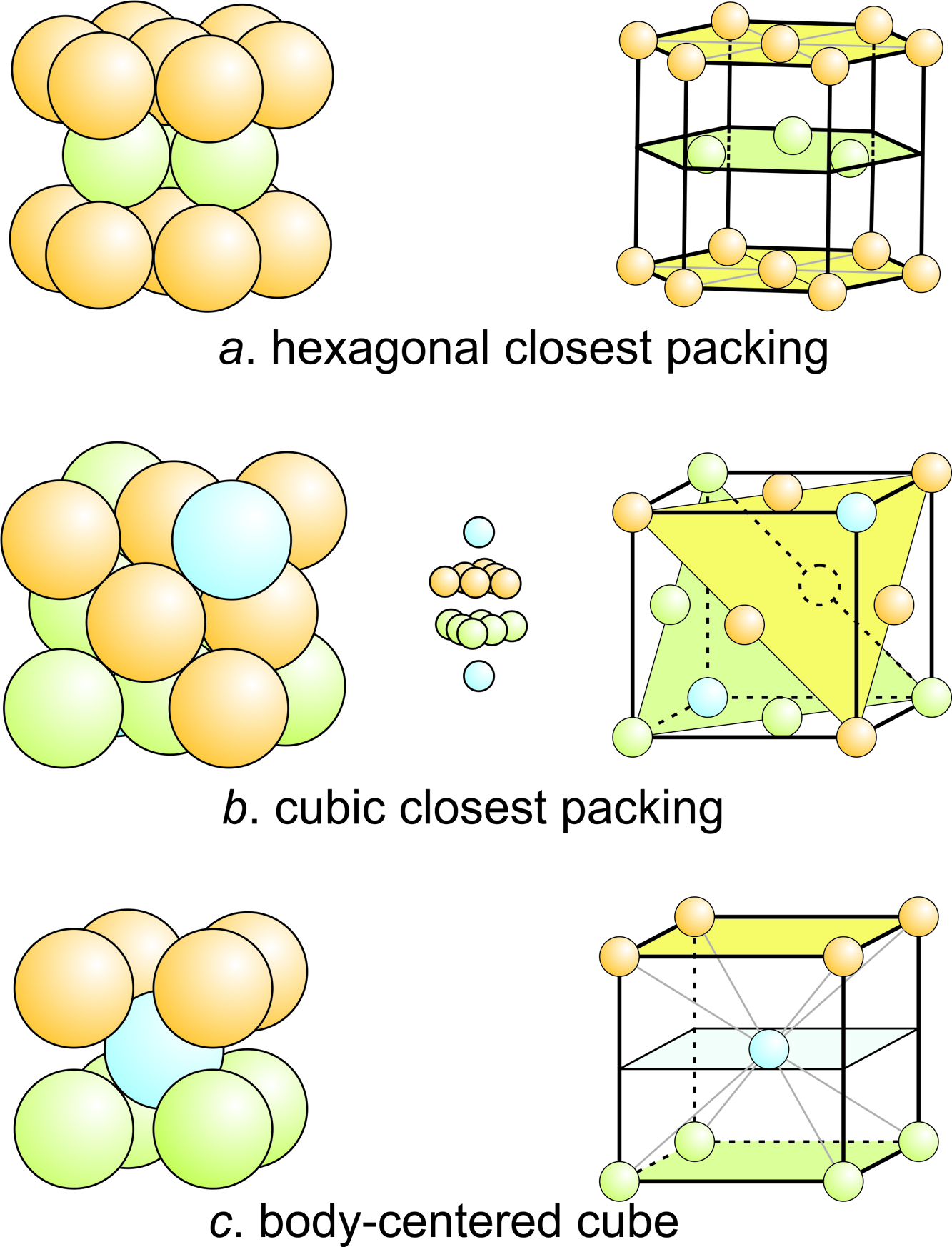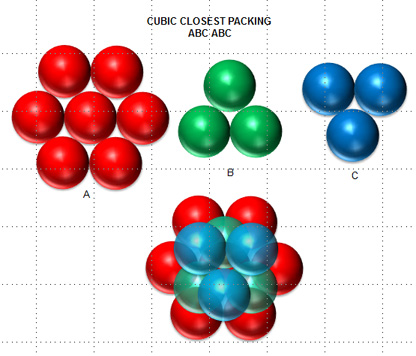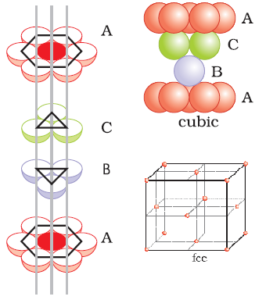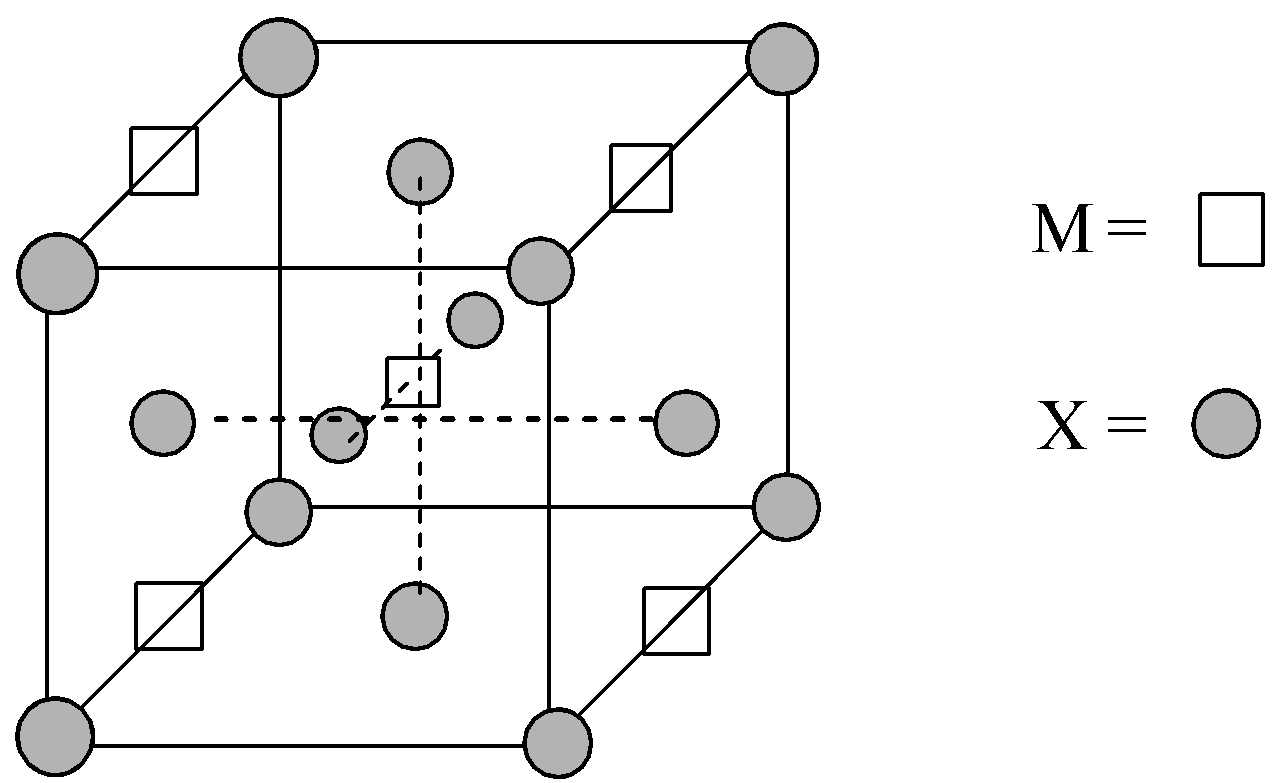
A compound ${M_p}{X_q}$ has cubic-close packing (c.c.p) arrangement of X. Its unit-cell structure is shown below. The empirical formula of the compound is:\n \n \n \n \n A) $MX$ B) $M{X_2}$ C) ${

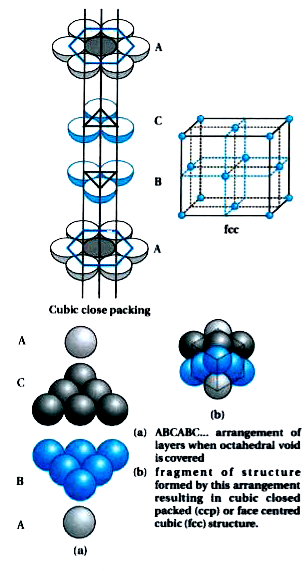
Statement: The cubic close - packed structure is based on an fcc unit cell.State whether the given statement is true or false